So for this experiment we used materials that are around the class which are:
Cardboard
Pennies
Zinc
Vinegar
Foil
LED light
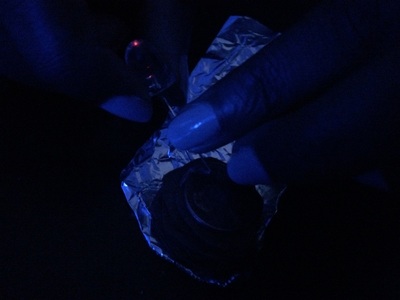
So for this we took 6 pennies and 10 round shaped pieces of cardboard and a beaker with vinegar. We took sand paper and used it to scrape one side of the pennies to remove the copper on that side. After all the pennies were done we took the cardboard pieces and dropped them vinegar. We padded zinc on top of tin foil and stacked cardboard and pennies one after another. After stacking we took a LED light and put the positive side on the tin foil and the negative on the penny that was on the stack.
The science behind is a little confusing but batteries are created to move electrons to one another. The current of electrons is going through the electrolyte and through the positive and negative charge. Zinc and the pennies scraped side is the negative charge. And then our positive charge is the rest of the penny with the copper. The cardboard put in vinegar are electrolytes. Without electrolytes its not able to carry electrical energy to help the electricity of the battery. The zinc and copper become the positive and negative electrons creating a circuit when you add the LED light.
We ended up getting great results from our battery. Overall this experiment was really fun and I learned a lot about elements having different attractions to electrons. I wouldn't mind learning more about this in the future I think its a interesting subject to get into. My group worked really great and from that we got results that we were looking for. Having no troubles was really nice and having everything go the way you want them always makes a lab 10 times better.
Cardboard
Pennies
Zinc
Vinegar
Foil
LED light
So for this we took 6 pennies and 10 round shaped pieces of cardboard and a beaker with vinegar. We took sand paper and used it to scrape one side of the pennies to remove the copper on that side. After all the pennies were done we took the cardboard pieces and dropped them vinegar. We padded zinc on top of tin foil and stacked cardboard and pennies one after another. After stacking we took a LED light and put the positive side on the tin foil and the negative on the penny that was on the stack.
The science behind is a little confusing but batteries are created to move electrons to one another. The current of electrons is going through the electrolyte and through the positive and negative charge. Zinc and the pennies scraped side is the negative charge. And then our positive charge is the rest of the penny with the copper. The cardboard put in vinegar are electrolytes. Without electrolytes its not able to carry electrical energy to help the electricity of the battery. The zinc and copper become the positive and negative electrons creating a circuit when you add the LED light.
We ended up getting great results from our battery. Overall this experiment was really fun and I learned a lot about elements having different attractions to electrons. I wouldn't mind learning more about this in the future I think its a interesting subject to get into. My group worked really great and from that we got results that we were looking for. Having no troubles was really nice and having everything go the way you want them always makes a lab 10 times better.


 RSS Feed
RSS Feed
