For this worksheet we learned about converting grams into moles and converting moles into grams. This worksheet was very useful because it more into the chemisty about chemistry if that makes any sense. After a few examples this worksheet became easier and I understood more about converting and what is really behind it.
|
I had trouble on the top part on this worksheet because I was confused if I had to put "ide" after the name of the element or not.
This worksheet was pretty easy after being showed an example I quickly understood how to do it and had really no trouble.
So for this lab we deiced to put a egg in a cup and add vinegar to it to watch the reaction.
The science behind this experiment was pretty straight forward its used to witness the importance of calcium for healthy teeth and bones. And how our bones and teeth become after the egg is put to testing. The vinegar reacts with the eggshells calcium. Because the vinegar is a mild acid. The eggshells solid calcium carbonate crystal makes and holds the shell together. When it hits soak in the vinegar, the acid breaks up the calcium. When is breaks it up the shell then begins to free itself and floats above the egg. Then leaving the egg to be rubbery. The steps for this were also really simple place the egg in glass make sure its empty, pour the vinegar inside covering the egg all the way, wait several days and take the egg out of the vinegar. We have to wait out a couple of days and make sure the egg is not disturbed, so you can get the results you will want. We used
Safety is a really big part in chemistry but for this project there really isn't anything harmful in the beginning since we are dealing with an egg and vinegar. But throughout the project there could be a chemical exposure with the vinegar and egg so we will used gloves at all times so we wouldn't catch anything. Over all this lab went great, I noticed the eggs were getting a weird shape and off color from the original and the smell it was giving was bad but those are just some steps it goes through over time and whats supposed to happen in the process of this lab. My partner and I worked worked really well and fast and got all of our work dine fast and safe I wouldn't change anything different. This lab was really interesting view the type pf reaction it gives and you really observe when you pay attention to everything going on. So for this experiment we used materials that are around the class which are:
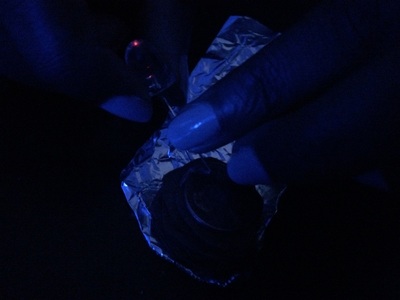
Cardboard Pennies Zinc Vinegar Foil LED light So for this we took 6 pennies and 10 round shaped pieces of cardboard and a beaker with vinegar. We took sand paper and used it to scrape one side of the pennies to remove the copper on that side. After all the pennies were done we took the cardboard pieces and dropped them vinegar. We padded zinc on top of tin foil and stacked cardboard and pennies one after another. After stacking we took a LED light and put the positive side on the tin foil and the negative on the penny that was on the stack. The science behind is a little confusing but batteries are created to move electrons to one another. The current of electrons is going through the electrolyte and through the positive and negative charge. Zinc and the pennies scraped side is the negative charge. And then our positive charge is the rest of the penny with the copper. The cardboard put in vinegar are electrolytes. Without electrolytes its not able to carry electrical energy to help the electricity of the battery. The zinc and copper become the positive and negative electrons creating a circuit when you add the LED light. We ended up getting great results from our battery. Overall this experiment was really fun and I learned a lot about elements having different attractions to electrons. I wouldn't mind learning more about this in the future I think its a interesting subject to get into. My group worked really great and from that we got results that we were looking for. Having no troubles was really nice and having everything go the way you want them always makes a lab 10 times better. So for this lab what we did was to create waterproof sand. It was something new brought to us and I like trying new things so i was really interested when Andrew told us about this and I was really to see what our outcome was going to be like at the end and if this could actually be pulled off.
Tiyonna Clark & Fernando Saucedo Lightning Experiment This worksheet was about ions and different type of elements. An ion is an element with a positive or negative net charge.The two Ions are a cation and a anion.I then showed electron dot structures showing the outer shell putting two electrons together.
|
Archives
May 2015
Categories |












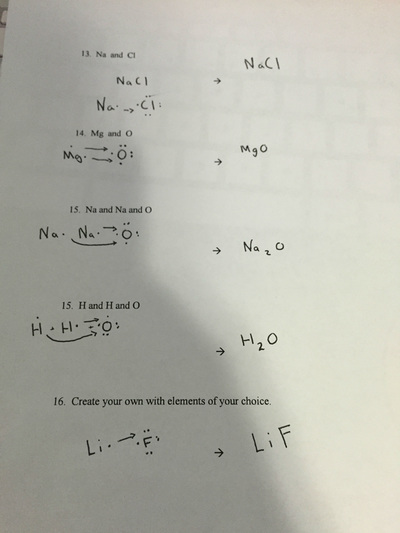

 RSS Feed
RSS Feed
